
This means that the negative charge on an electron perfectly balances the positive charge on the proton.
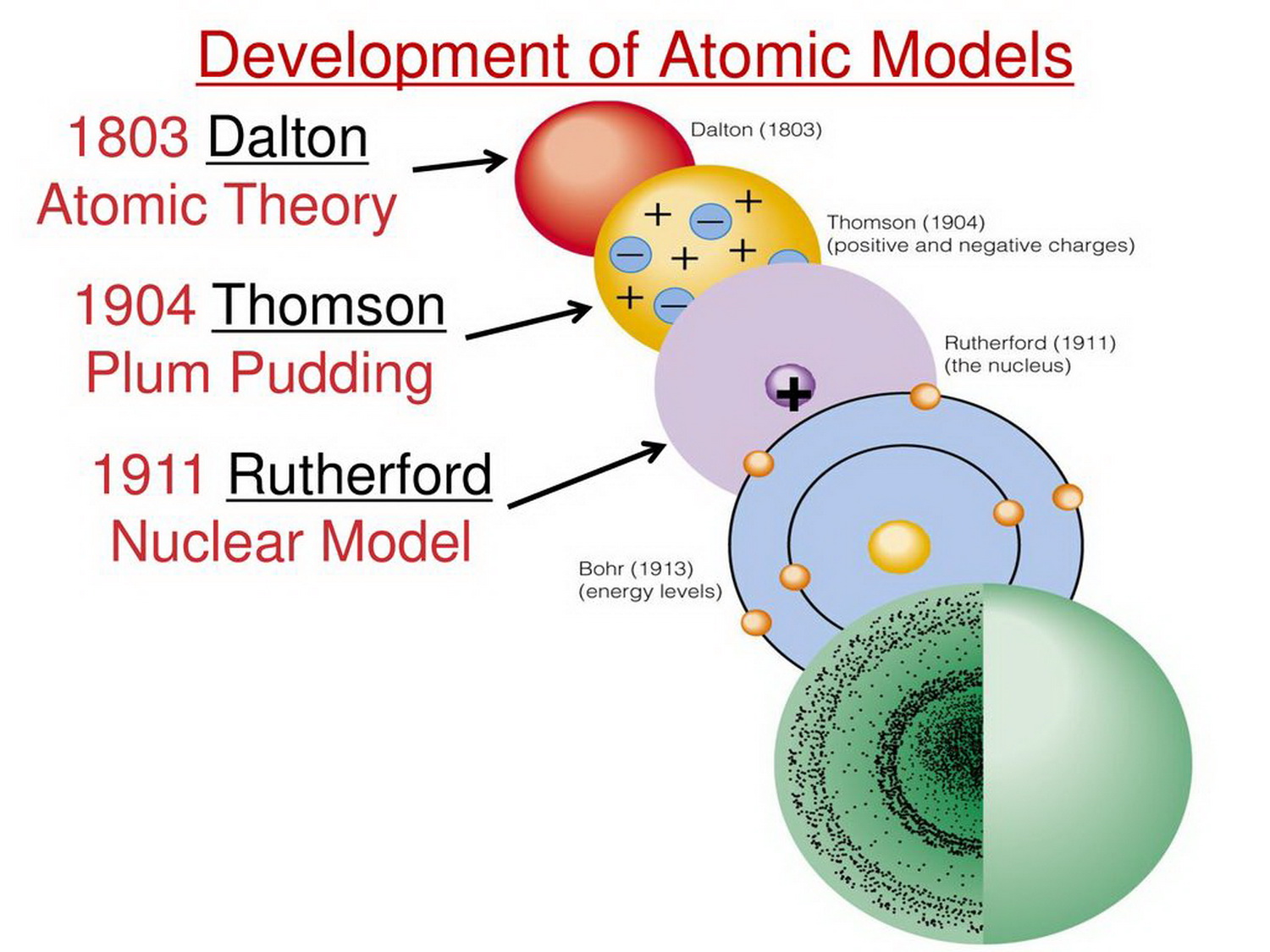
The existence of isotopes illustrates this phenomena. All atoms of an element are not identical. We know now that: An atom is composed of smaller particles (electrons, protons, and neutrons). Negative and positive charges of equal magnitude cancel each other out. Daltons atomic theory has been largely accepted by the scientific community, with the exception of three changes. These ancient philosophers speculated that the earth was made up of different combinations of basic substances, or elements. It must be added, atomism was one of a number of competing theories on the nature of matter. Neutral atoms have the same number of electrons as they have protons, so their overall charge is zero. Atomic theory began as a philosophical concept in ancient Greece and India. We will discuss isotopes and their symbols more in section 4.5 of this chapter. Atoms of the same element (i.e., atoms with the same number of protons) with different numbers of neutrons are called isotopes. The sum of the number of protons and neutrons in the nucleus is called the mass number of the isotope. Each element has its own characteristic atomic number.Ītoms of the same element can have different numbers of neutrons, however. He shared the 1933 Nobel Prize for Physics with British physicist P.A.M. He also made significant contributions to nearly all branches of science and philosophy. Thus, hydrogen has an atomic number of 1, while iron has an atomic number of 26. Erwin Schrodinger, Austrian theoretical physicist who contributed to the wave theory of matter and to other fundamentals of quantum mechanics. The number of protons in an atom is the atomic number of the element (Z).

This number of protons is so important to the identity of an atom that it is called the atomic number. All atoms of hydrogen have one and only one proton in the nucleus all atoms of iron have 26 protons in the nucleus. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Each element has its own atomic number, which is equal to the number of protons in its nucleus. Atoms themselves are composed of protons, neutrons, and electrons. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. \): The Structure of the Atom.Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus.
